|
7 Approximately 0.3 percent of 48 million blood donors who reported no high-risk behavior for HIV-1 infection at the time of donation had repeatedly reactive enzyme immunoassays, 8, 9 but the majority of these donors have subsequently tested negative on Western blotting. 5 This testing sequence has revealed a low seroprevalence of Western blot-positive donations (0.018 percent of 15 million blood donations to the American Red Cross between April 1985 and May 1988) 6 and has been effective in reducing the risk of transmission of HIV-1 infection by blood transfusion. 4 This assay typically reveals six to nine characteristic bands if antibodies to HIV-1 proteins of different molecular weights are present and no bands if such antibodies are absent. 2 To verify the presence of HIV-1 antibody in a repeatedly reactive sample, the Western blot assay 3 has most often been used as a confirmatory test in the United States. 1 The blood from donors who have been found to be repeatedly reactive on an enzyme immunoassay for HIV-1 antibody has not been used in transfusion or manufactured into other products capable of transmitting infectious agents. LICENSED enzyme immunoassay kits for screening the blood of volunteer blood donors for antibody to the human immunodeficiency virus type 1 (HIV-1) have been available since March 1985. We conclude that persons at low risk for HIV infection who have persistent indeterminate HIV-1 Western blots are rarely if ever infected with HIV-1 or HIV-2. No donor met the criteria for a positive Western blot assay for HIV-1, and none had evidence of HIV-1 or HIV-2 infection on culture or by any other test. In 91 subjects (92 percent) the Western blot results were still indeterminate, whereas in 8 they were negative. After a median of 14 months (range, 1 to 30) from the time of the initial test, 65 subjects (66 percent) were still repeatedly reactive for HIV-1 antibody on at least one immunoassay. Of the 99 blood donors, 98 reported no risk factors for HIV-1 infection 1 donor had used intravenous drugs. Coded blood specimens were tested again for HIV-1 antibody (by two different enzyme immunoassays, a Western blot assay and a radioimmunoprecipitation assay) and for HIV-2 antibody by enzyme immunoassay, for HIV-1 by the serum antigen test, for HIV-1 by culture, for human T-cell leukemia virus Type I or II antibody by enzyme immunoassay, and for sequences of HIV DNA by the polymerase chain reaction. The subjects were interviewed about HIV risk factors. To determine whether apparently healthy persons who have had repeatedly reactive enzyme immunoassays and an indeterminate Western blot assay for antibody to the human immunodeficiency virus type 1 (HIV-1) are infected with HIV-1 or HIV-2, we studied 99 such volunteer blood donors in a low-risk area of the country. The most trusted, influential source of new medical knowledge and clinical best practices in the world. Information and tools for librarians about site license offerings. Valuable tools for building a rewarding career in health care. The authorized source of trusted medical research and education for the Chinese-language medical community. The most advanced way to teach, practice, and assess clinical reasoning skills. 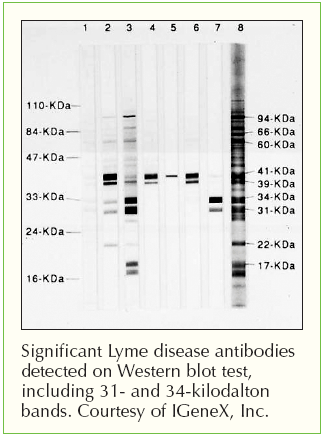
Information, resources, and support needed to approach rotations - and life as a resident. The most effective and engaging way for clinicians to learn, improve their practice, and prepare for board exams. NEW! Peer-reviewed journal featuring in-depth articles to accelerate the transformation of health care delivery.Ĭoncise summaries and expert physician commentary that busy clinicians need to enhance patient care. NEW! A digital journal for innovative original research and fresh, bold ideas in clinical trial design and clinical decision-making.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
